9 May 2014
How neurogenesis finds its place in a hardwired sensory system
Livio Oboti1 and Paolo Peretto2
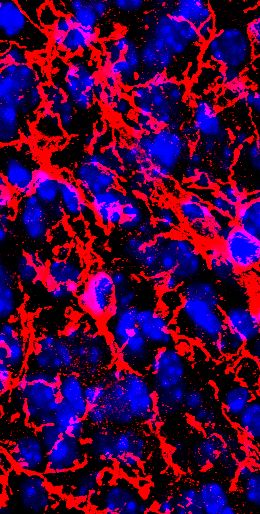
So far most studies on adult neurogenesis aimed to unravel mechanisms and molecules regulating the integration of newly generated neurons in the mature brain parenchyma. The exceedingly abundant amount of results that followed, rather than being beneficial in the perspective of brain repair, provided a clear evidence that adult neurogenesis constitutes a necessary feature to the correct functioning of the hosting brain regions. In particular, the rodent olfactory system represents a privileged model to study how neuronal plasticity and neurogenesis interact with sensory functions. Until recently, the vomeronasal system (VNS) has been commonly described as being specialized in the detection of innate chemosignals.
Accordingly, its circuitry has been considered necessarily stable, if not hard-wired, in order to allow stereotyped behavioral responses. However, both first and second order projections of the rodent VNS continuously change their synaptic connectivity due to ongoing postnatal and adult neurogenesis. How the functional integrity of a neuronal circuit is maintained while newborn neurons are continuously added—or lost—is a fundamental question for both basic and applied neuroscience. The VNS is proposed as an alternative model to answer such question. Hereby the underlying motivations will be reviewed. [read full article]
1Children's National Health System, Center for Neuroscience Research, Washington, DC, USA
2Department of Life Sciences and Systems Biology, Neuroscience Institute Cavalieri Ottolenghi, University of Torino, Orbassano, Italy
23 May 2014
Beyond cell replacement: unresolved roles of NG2-expressing progenitors
Enrica Boda and Annalisa Buffo*
NG2-expressing parenchymal precursors (NG2+p) serve as primary source of myelinating oligodendrocytes in both the developing and adultCentral Nervous System (CNS). However, their abundance, limited differentiation potential at adult stages along with stereotypic reaction to injury independent of the extent of myelin loss suggest that NG2+p exert functions additional to myelin production. In support of this view, NG2+p express a complex battery of molecules known to exert neuromodulatory and neuroprotective functions.
Further, they establish intimate physical associations with the other CNS cell types, receive functional synaptic contacts and possess ion channels apt to constantly sense the electrical activity of surrounding neurons. These latter features could endow NG2+p with the capability to affect neuronal functions with potential homeostatic outcomes. Here we summarize and discuss current evidence favoring the view that NG2+p can participate in circuit formation, modulate neuronal activity and survival in the healthy and injured CNS, and propose perspectives for studies that may complete our understanding of NG2+p roles in physiology and pathology. [read full article]
*Department of Neuroscience Rita Levi-Montalcini, Neuroscience Institute Cavalieri Ottolenghi, University of Turin, Turin, Italy
Agenda
Area Ricercatori
Guarda il video
GiovedìScienza racconta la ricerca al NICO
Vivere per sempre.
Una popolazione sempre più longeva, i suoi problemi e le risposte della ricerca
Hai perso la diretta? Guarda ora il video di GiovedìScienza al NICO: una puntata in diretta dai nostri laboratori dedicata alla ricerca sull'invecchiamento.